Benchmark Dose Tools
Predicting chemical dose and estimating the quantitative relationship between this dose and a response are critical components of risk assessment.
EPA's Benchmark Dose Software (BMDS) provides easy access to numerous mathematical models. These models help risk assessors estimate the quantitative relationship between a chemical dose and a response.
EPA’s BMD tools include BMDS (Online, Desktop, pybmds), and Categorical Regression (CatReg).
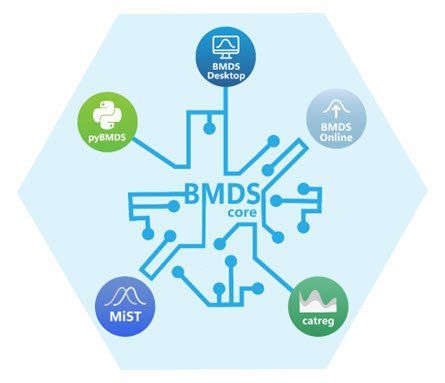
-
About Benchmark Dose Modeling
Learn more about Benchmark Dose Modeling and how it is used in risk assessment.
-
Benchmark Dose Tools Suite
Explore the suite of BMDS tools that support benchmark dose modeling.
-
BMDS Training and Resources
View training videos and aids that go beyond the software documentation.
