Basic Ozone Layer Science
I. The Ozone Layer
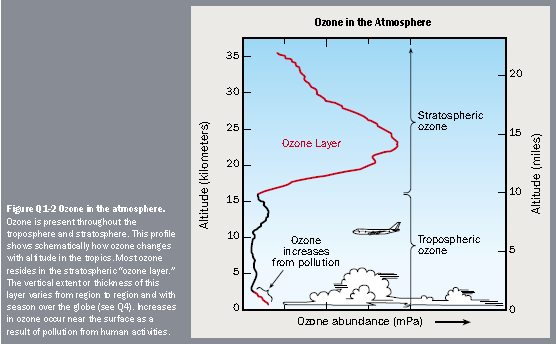
The Earth's atmosphere is composed of several layers. The lowest layer, the troposphereThe region of the atmosphere closest to the Earth. The troposphere extends from the surface up to about 10 km in altitude, although this height varies with latitude. Almost all weather takes place in the troposphere. Mt. Everest, the highest mountain on Earth, is only 8.8 km high. Temperatures decrease with altitude in the troposphere. As warm air rises, it cools, falling back to Earth. This process, known as convection, means there are huge air movements that mix the troposphere very efficiently., extends from the Earth's surface up to about 6 miles or 10 kilometers (km) in altitude. Virtually all human activities occur in the troposphere. Mt. Everest, the tallest mountain on the planet, is only about 5.6 miles (9 km) high. The next layer, the stratosphereThe region of the atmosphere above the troposphere. The stratosphere extends from about 10km to about 50km in altitude. Commercial airlines fly in the lower stratosphere. The stratosphere gets warmer at higher altitudes. In fact, this warming is caused by ozone absorbing ultraviolet radiation. Warm air remains in the upper stratosphere, and cool air remains lower, so there is much less vertical mixing in this region than in the troposphere., continues from 6 miles (10 km) to about 31 miles (50 km). Most commercial airplanes fly in the lower part of the stratosphere.
Most atmospheric ozone is concentrated in a layer in the stratosphere, about 9 to 18 miles (15 to 30 km) above the Earth's surface (see the figure). Ozone is a molecule that contains three oxygen atoms. At any given time, ozone molecules are constantly formed and destroyed in the stratosphere. The total amount has remained relatively stable during the decades that it has been measured.
Scientists have established records spanning several decades that detail normal ozone levels during natural cycles. Ozone concentrations in the atmosphere vary naturally with sunspots, seasons, and latitude. These processes are well understood and predictable. Each natural reduction in ozone levels has been followed by a recovery. Beginning in the 1970s, however, scientific evidence showed that the ozone shield was being depleted well beyond natural processes.
II. Ozone Depletion
When chlorine and bromine atoms come into contact with ozone in the stratosphere, they destroy ozone molecules. One chlorine atom can destroy over 100,000 ozone molecules before it is removed from the stratosphere. Ozone can be destroyed more quickly than it is naturally created.
Some natural processes, such as large volcanic eruptions, can have an indirect effect on ozone levels. For example, Mt. Pinatubo's 1991 eruption did not increase stratospheric chlorine concentrations, but it did produce large amounts of tiny particles called aerosolsSmall particles or liquid droplets in the atmosphere that can absorb or reflect sunlight depending on their composition. (different from consumer products also known as aerosols). These aerosols increase chlorine's effectiveness at destroying ozone. The aerosols in the stratosphere create a surface on which CFC-based chlorine can destroy ozone. However, the effect from volcanoes is short-lived.
Not all chlorine and bromine sources contribute to ozone layer depletion. For example, researchers have found that chlorine from swimming pools, industrial plants, sea salt, and volcanoes does not reach the stratosphere. In contrast, ODS are very stable and do not dissolve in rain. Thus, there are no natural processes that remove the ODS from the lower atmosphere.
One example of ozone depletion is the annual ozone "hole" over Antarctica that has occurred during the Antarctic spring since the early 1980s. This is not really a hole through the ozone layer, but rather a large area of the stratosphere with extremely low amounts of ozone.
Ozone depletion is not limited to the area over the South Pole. Research has shown that ozone depletion occurs over the latitudes that include North America, Europe, Asia, and much of Africa, Australia, and South America. More information about the global extent of ozone depletion can be found in the Scientific Assessment of Ozone Depletion: 2022 developed by the United Nations Environment Programme.
