Metals
Overview

These metals can reach water bodies when they are released into the air, water and soil. Unlike sediment and nutrient impairments, there is often no visible evidence of metals contamination.
While some metals are essential as nutrients, all metals can be toxic at some level. Some metals are toxic in minute amounts. Impairments result when metals are biologically available at toxic concentrations affecting the survival, reproduction and behavior of aquatic organisms.
Checklist of Sources, Site Evidence and Biological Effects
This module addresses water column contamination by metals and metalloids that commonly cause toxic effects. These include arsenic, cadmium, chromium, copper, lead, inorganic mercury, nickel, selenium and zinc. Organic mercury and tributyl tins are special forms of metals that are beyond the scope of this module.
Metals should be a candidate cause when human sources and activities, site observations or observed biological effects support portions of the source-to-impairment pathways (Figure 1). This conceptual diagram for metals and some of the other information also may be useful in Step 3: Evaluate Data from the Case.
The checklist below will help you identify key data and information useful for determining whether to include metals among your candidate causes. The list is intended to guide you in collecting evidence to support, weaken or eliminate metals as a candidate cause.
For more information on specific entries, click on checklist headings or go to the When to List tab.
Sources and Activities
- Mines and smelters
- Firing ranges
- Municipal waste treatment outfalls
- Industrial point sources
- Urban runoff
- Landfills
- Junkyards
Site Evidence
- Blue, orange or yellow precipitate in water
- Site data for metals
- Site chemistry favoring metals bioavailability
Biological Effects
- Kills of aquatic life
- Mucous streaming from gills
- Gill damage
- Blue stomachs (molybdenum)
- Spinal abnormalities (calcium analogs)
- Blackened tails
- Replacement of metals-sensitive species with tolerant species
- Unspecified toxic chemicals
- Ionic strength
Consider other causes with similar evidence when considering metals as a candidate cause:
When to List
- Sources and Activities that Suggest Listing Metals as a Candidate Cause
- Site Evidence that Suggests Listing Metals as a Candidate Cause
- Biological Effects that Suggest Listing Metals as a Candidate Cause
- Site Evidence that Supports Excluding Metals as a Candidate Cause
Sources and Activities that Suggest Listing Metals as a Candidate Cause
One reason to list metals as a candidate cause is the presence of metal sources or other evidence of metals in a stream or watershed. When identifying sources of metals, consider both non-point and point sources.

Disturbance and redistribution of metals-contaminated sediments by dredging can result in repartitioning of metals into the water column. It is important to consider that sediment may contain legacy metals-contamination from past land uses.
The relative distribution of urbanized areas contributing non-point metals and other toxicants within a watershed can be identified using U.S. EPA's My WATERS Mapper.
Industrial sources of atmospheric releases of metals can be identified by querying the Toxics Release Inventory.
Information from the TRI database should only be used to identify which metals may be present. Stack releases in an area do not necessarily result in actual exposures due to differing release-specific fate and transport pathways. In addition, location metadata might identify the company headquarters rather than the point of release, so confirming the location of actual release is recommended.
Point source contributions of metals to surface waters include releases by different industries and by wastewater treatment facilities. Point source contributions from water treatment facilities may be identified through permits granted under the National Pollutant Discharge Elimination System.
Other evidence of metals in the watershed or site area may be identified by examining:
- State or tribal databases
- U.S. EPA's STOrage and RETrieval (STORET) and Water Quality eXchange (WQX)
- U.S. Geological Survey's National Water-Quality Assessment Program (NAWQA)
- U.S. EPA's Environmental Monitoring and Assessment Program (EMAP)
Site Evidence that Suggests Listing Metals as a Candidate Cause
One also may list metals as a candidate cause when they have been measured at the site. The availability of data on metals concentrations in biota, sediments or water suggests that metals co-occur with the impairment.

When listing metals, also consider how site water chemistry conditions influence metal bioavailability and toxicity. This will be important in later stages of the assessment. The Issue Paper on the Environmental Chemistry of Metals reviews important environmental chemistry factors influencing metal bioavailability.
- Issue Paper on the Environmental Chemistry of Metals (Langmuir et al. 2004)
- Issue Paper on the Ecological Effects of Metals (Kapustka et al. 2004)
The fraction of metals present in the water column as biologically available free metal ions is particularly important. High free metal ion concentrations are toxic because they compete with nutrient cations (e.g., calcium, potassium, magnesium) for binding sites located on chloride cells of gill epithelia (biotic ligands). This impairs gill respiratory function and the ability to regulate blood pH and ion concentrations. Metals are not available for gill binding when they exist as organic compounds or are bound to sulfates, organic acids, other anions or negatively charged particles (abiotic ligands).
Data for individual ions may be used in calculating ion balance while alkalinity, hardness, and dissolved organic carbon are common aggregate measures of abiotic ligands. Low pH of the water favors metal solubility, increasing free metal ion content, particularly when abiotic ligands are not abundant. Taken together, the concentration of metal ions relative to nutrient ions, the abundance of abiotic ligands and pH determine the biologically available fraction and toxicity of metals in water (Di Toro et al. 2001).
Biological Effects that Suggest Listing Metals as a Candidate Cause
- Mucous streaming from fish gills, due to gill injury or impaired ionoregulation (Hunn and Schnick 1990)
- Degeneration of caudal chromophores resulting in black tails in fish (Bengtsson and Larsson 1986, Sippel et al. 1983)
- Skeletal deformities and impaired growth and development, for example due to metals acting as analogs for calcium and sulfur (Sorensen 1991)
- Abnormal organ appearances or parasite loads (Sures 2001) (although organ pathologies are seldom available)
- Reduced abundance of metal-sensitive invertebrate taxa, such as mayflies (Pollard and Yuan 2005)
- Increased abundance of relatively metal-tolerant taxa such as caddisflies and many stoneflies (Clements et al. 1992)
Metals alter communities because species differ in sensitivity. Different taxa have different chloride cell densities on their gills. This influences their vulnerability to effects on respiration and the regulation of blood pH and ion concentrations (Di Toro et al. 2001). Taxa also have different metabolic mechanisms for detoxifying, sequestering and excreting metals. Some taxa can acclimate to chronic metals exposure by increasing the capacity of these metabolic mechanisms. Metals accumulated by organisms enter the food chain and can contribute to toxic effects through dietary exposures.
Site Evidence that Supports Excluding Metals as a Candidate Cause
We strongly caution against using benchmarks of effects (e.g., water quality criteria) as evidence for excluding metals from your initial list of candidate causes. Different species have different metal requirements and different sites have different naturally occurring levels of metals.
Ways to Measure
Unlike certain water quality parameters (e.g., pH, temperature, conductivity), metals are not commonly measured on site. Water, sediment and tissue samples must be returned to the laboratory for processing and analysis. Details on how to obtain uncompromised water samples can be found in:
- U.S. EPA Method 1669: Sampling Ambient Water for Trace Metals at EPA Water Quality Criteria Levels (42 pp, 114 K, About PDF)
Information on appropriate sample handling and methods for analysis can be found in:
- U.S. EPA's Methods for the Determination of Metals in Environmental Samples(308 pp, 7 Mb, About PDF)
- U.S. EPA's Methods for the Determination of Metals in Environmental Samples, Supplement 1(260 pp, 700 K, About PDF)
Water samples often are filtered to measure dissolved metal concentrations and acidified to keep metals in solution. Operationally, a metal is considered to be dissolved if it remains in solution after filtration through a 0.45 micromolar (uM) filter. Suspended metals are those that remain on the 0.45 μM filter. “Acid extractable metals” refers to the concentration of metals after acidification of an unfiltered sample. It is used primarily to estimate drinking water exposures rather than aquatic life exposures.
Biota may be prepared as whole organisms, or individual organs may be dissected out and analyzed. Biological samples are often dried to a constant mass before analysis and data are reported as “dry mass”. Percent moisture [i.e., (1-mass after drying / mass before drying) *100] for these samples may be used to back calculate the original tissue concentration [i.e., 1-(%moisture/100)*[metal]. Samples may be analyzed fresh and reported as “fresh” or “wet” mass. Dry mass may be calculated using the percent moisture reported for a sub-sample. Preparation of biota and sediment for analysis involves dissolution of organic materials through digestion in nitric acid followed by hydrogen peroxide. Sometimes perflouric acid is used when digesting sediment to ensure that mineral compounds dissolve and to release metals bound in crystalline matrices.
Conceptual Diagrams
About Conceptual Diagrams
Conceptual diagrams are used to describe hypothesized relationships among sources, stressors and biotic responses within aquatic systems.
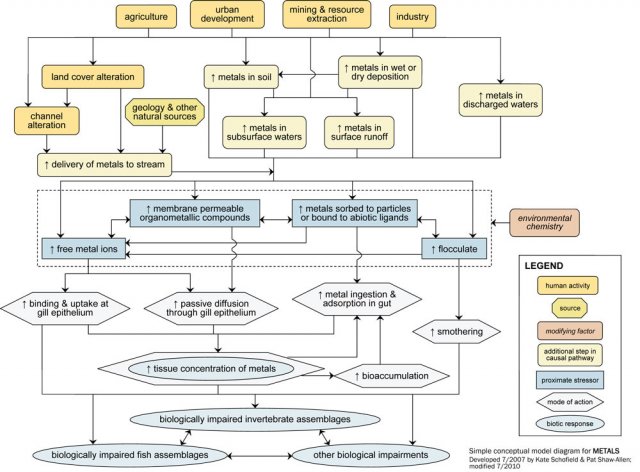
Simple Conceptual Model Diagram
Metals enter surface waters from point and non-point sources resulting from a variety of human land use practices. A point source is a direct input from a discrete source, usually an outfall discharging effluent to surface water. Non-point sources include atmospheric emissions and land uses which contaminate soil with metals through the extraction, refinement and use of fossil fuel and mineral resources. Atmospheric emissions can deposit onto surface waters or land surfaces. Metals on land and in soil can infiltrate into subsurface waters (e.g., groundwater) or wash into streams during storms.
Naturally metals-enriched regions become non-point sources when land cover alterations expose rock and soil to erosion. These pathways also apply to areas with legacy contamination, or historical sources (see Figure 2). Legacy contamination can include soil contamination from arsenical pesticide residues and long term industrial or urban land use. Land cover alterations which reduce watershed and riparian vegetation (e.g., due to agriculture, forestry, residential and commercial development) decrease infiltration and increase the volume and velocity of stormwater runoff. Channel alteration and increased flow can incise channels and reduce bank stability, resulting in bank and channel erosion and redistribution of metals-enriched substrates. More detailed information on metals sources and factors conveying metals to surface waters can be found in Marsalek et al. (2006).
Once in water, the bioavailability and toxicity of a metal is determined by its speciation, which is itself largely determined by several environmental parameters (e.g., pH, temperature, redox potential, ionic strength, presence of methylating microbes, and the availability of binding sites). Based on these parameters, free metal ions may precipitate as flocculates, form complexes with ligands (i.e., biotic or abiotic binding sites), become transformed to organometallic compounds (e.g., methylation in mercury), or sorb to solid particles. For more details on metals speciation, see Langmuir et al. (2004).
The metal species of primary toxicological concern are free ions. For many metals, acute toxicity results from binding of free metal ions onto chloride cells of the gill epithelium, disrupting osmoregulation and leading to mortality (Di Toro et al. 2001). Certain metal forms may diffuse into the gill epithelium, but these routes of absorption are considered minor relative to free ion binding. Toxicity associated with dietary bioaccumulation of metals and, for some metals, biomagnification within food webs can occur when there are sustained exposures in habitats with persistent contamination. Acid mine drainage can present a physical cause of impairment when drainage water mixes with the higher pH water of a receiving stream and metal hydroxides precipitate. The flocculate that results coats the stream bed and can smother organisms and their benthic habitat.
The physiological mechanisms of metal toxicity may translate into a broad spectrum of organism-level effects, ranging from altered behavior to outright lethality. Biotic responses are both taxa- and metal-specific, and may include increased occurrence of deformities or increased mucous secretion in fish. Differences in susceptibility to metals toxicity can restructure communities. For more details on the ecological effects of metals, see Kapustka et al. (2004).
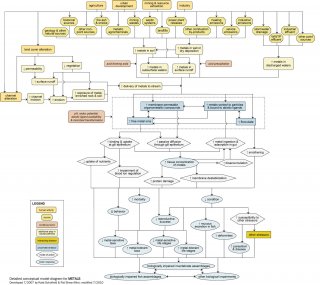
Detailed Conceptual Model Diagram
The following conceptual diagram (see Figure 3) describes the pathways by which point and non-point sources may contribute metals to surface waters, how water chemistry influences bioavailability, and how biologically available metals may impair aquatic life. It illustrates linkages between metals-related stressors (middle of diagram), the human activities and sources that can increase those stressors (top of diagram), and the biological responses that can result (bottom of diagram).
In some cases, additional steps leading from sources to stressors, modes of action leading from stressors to responses, and other modifying factors also are shown. This narrative generally follows the diagram top to bottom, then left to right.
Linking Sources to Proximate Stressors
Metals enter surface waters by means of point and non-point sources (in tan octagons). A point source is a direct release to water from a discrete source, usually an outfall discharging effluent to surface water. Non-point sources include atmospheric emissions and land uses which contaminate soil with metals. Naturally metals-enriched regions such as the Colorado Mineral Belt become non-point sources when land cover alterations (e.g., devegetation due to agricultural or mining practices) expose rock and soil, which erode into surface waters. These pathways also apply to areas with legacy contamination, such as soil contamination from arsenical pesticide residues or long term industrial or urban land use. Reduced water transpiration due to devegetation and reduced infiltration due to the increased impervious surface cover (compacted soil, roofs, parking lots, and roads) increases the volume and velocity of stormwater runoff entering surface waters. Accelerated flow can incise channels, reducing bank stability and increasing bank and channel erosion. Bank and channel erosion also may be increased by livestock grazing and trampling. Stormwater turbulence or intentional dredging can re-suspend sediments, which may allow sediment-associated metals to partition into the water column, or transport contaminated sediment into previously uncontaminated areas.
Metals entering the atmosphere from tailpipe and stack emissions are precipitated onto land or directly onto water. Episodic, pulsed exposures occur when metals precipitated onto land are washed into surface waters during storms. Smaller runoff events may result from activities such as washing cars or watering lawns and landscaping. The severity of episodic exposures is related to the amount of dry deposition built up in the period between events, saturation levels of non-impervious areas, and the volume of water discharged. The highest levels occur early in the runoff period, or in the “first flush.” Metal mobility can be increased by acid rain or soils with acid-forming parent material, fertilizers, tailings or other amendments. More gradual releases occur over periods of snowmelt which can contribute metals to both soil moisture and direct runoff. More detailed information on metals sources and factors conveying metals to surface waters can be found in Marsalek et al. (2006).
Waste from residential and commercial areas may be directed to septic systems, landfills or wastewater treatment plants. Septic systems and landfills can contribute to metals contamination via leachate entering subsurface waters (e.g., groundwater) and eventually surface waters. Wastewater treatment plants, on the other hand, discharge metals-contaminated effluent directly into surface waters. Industrial activities contributing metals to the environment include the storage, refinement, and combustion of fossil fuels and the manufacture of a variety of goods. Waste from industrial facilities may be treated on-site and discharged, stored on-site, or directed to off-site landfills or wastewater treatment plants. Leachate and surface runoff from on-site or off-site storage and disposal areas may transport metals to surface waters. Mining also may involve on-site storage and disposal of wastes, which may contribute metals to surface waters via similar pathways.
Linking Proximate Stressors to Biological Impairment
Once in water, the bioavailability and toxicity of a metal is determined by its partitioning and speciation, which are determined by several environmental parameters (e.g., pH, temperature, redox potential, ionic strength, presence of methylating microbes, and the availability of binding sites). Based on these parameters, free metal ions may precipitate, form complexes with ligands (i.e., biotic or abiotic binding sites), become transformed to organometallic compounds (e.g., methylation in mercury), or sorb to solid particles. For more details on metals speciation, see Langmuir et al. (2004).
The metal species of primary toxicological concern are free ions. For many metals, acute toxicity results from binding of free metal ions to chloride cells of the gill epithelium, disrupting osmoregulation and leading to mortality (Di Toro et al. 2001). Certain metal forms may diffuse into the gill epithelium, but these absorption routes are considered minor relative to free ion binding. Toxicity associated with dietary bioaccumulation of metals and, for some metals, biomagnification within food webs can occur when there are sustained exposures in habitats with persistent contamination; in addition, food source and feeding mode are key factors determining whether dietary bioaccumulation occurs. Acid mine drainage can present a physical cause of impairment when drainage water mixes with the higher pH water of a receiving stream and metal hydroxides precipitate. The flocculate that results can coat the streambed and smother benthic habitats and organisms.
Interactions with gill membranes can damage tissue and impair the ionoregulatory processes of chloride cells. Once within the organism, metal toxicity may occur by the substitution of nutrients with toxic analogs (e.g., Pb ↔ Ca), improper insertion into enzyme active sites (e.g., Se ↔ S), oxidative injury via free radical cycling, and nonspecific binding to nucleic acids and proteins. Analog substitution and nonspecific binding impair proper function of biomolecules, which in turn influences the physiological health of the organism. Impairment of proteins associated with organelle and cellular membranes affect membrane integrity, leading to breaches and impaired function. At the gill, impairment of chloride cells on the gill affects ion regulation. Internally, impairment of mitochondrial membranes cause leakage of oxyradicals generated via the electron transport pathway. Oxyradicals in turn can cause oxidative damage leading possibly to cell death.
The physiological mechanisms of metal toxicity may translate into a broad spectrum of organism-level effects, ranging from altered behavior (avoidance of contaminated areas, increased susceptibility to predation or reduced success of predators) to outright lethality. Responses detectable in biosurveys often are limited to increases in the relative abundance of metal-tolerant species, decreases in metal-sensitive species, and certain physical anomalies observed in fish (e.g., selenium-associated spinal deformities, blackened tails due to collapse of chromophores, excess mucous production due to gill irritation, and impaired ionoregulation). Examples of species often considered metal-tolerant include chironomids, caddisflies, small-bodied stoneflies, yellow perch and central stonerollers; examples of metal-sensitive species include bivalves, mayflies, and salmonids. However, it should be kept in mind that biotic responses are both taxa- and metal-specific. For more details on the ecological effects of metals, see Kapustka et al. (2004).
Literature Reviews
This section provides an annotated bibliography of literature reviews that relate biological effects to metals concentrations, to help you identify useful information for Step 4: Evaluate Data from Elsewhere.
Documents in the list below are included if they contain information on stressor intensities and biological responses in the field or if they compile field-relevant stressor-response information not yet covered in the ECOTOX database. For many metals, the U.S. EPA also has published water quality criteria documents.
- Beak International Incorporated (2002) Literature review of environmental toxicity of mercury, cadmium, selenium and antimony in metal mining effluents. Natural Resources Canada, The Mining Association of Canada, and Environment Canada.
This report characterizes the metal content and other properties of mine effluents and summarizes speciation (including Eh-pH diagrams), fate, transport and toxicity (acute laboratory tests). Includes information on mercury, cadmium, selenium and antimony concentrations in untreated and treated mine effluent,and a review table of acute toxicity in fish and invertebrates. Speciation figures and text are provided for mercury, cadmium, selenium and antimony.
- Borgmann U, Couillard Y, Doyle, Dixon DG (2005) Toxicity of sixty-three metals and metalloids to Hyalella azteca at two levels of water hardness. Environmental Toxicology and Chemistry 24(3):641-652.
This paper is not a literature review. It reports data from toxicity tests of chemicals in water and will eventually be included in the ECOTOX database. It is included here because it is an extensive work that examines the influence of water hardness on metal toxicity. We provide a data extraction (Borgmann et al. 2005 data) to facilitate data comparisons that are not possible in ECOTOX. In particular, we calculated relative breadth (mean divided by span of 95% confidence interval) to provide a weighted relative uncertainty in LC50 estimates to facilitate such comparisons.
Borgmann et al. 2005 Data (xls) (.xls file)
- Naimo TJ (1995) A review of the effects of heavy metals on freshwater mussels. Ecotoxicology 4:341-362.
This review characterizes metal concentrations in different water bodies and describes the distribution and metabolism of metals in freshwater mussels. Text on page 352 reviews data on chronic effects. Table 2 reports data for acute laboratory tests for cadmium, copper, nickel and zinc. These data also are in ECOTOX, but we suggest examining the original document to compare toxicity data and review how metals interact with biota at a metabolic level. The review also lists citations reviewing alternative causes (other than metals) of mussel decline.
- Rowe CL, Hopkins WA, Congdon JS (2002) Ecotoxicological implications of aquatic disposal of coal combustion residues in the United States: a review. Environmental Monitoring and Assessment 80:207-276.
This paper characterizes coal combustion residues, concentrations observed in associated water, sediment and biota, and toxicity to biota. Data for selenium are relatively abundant. Table VII lists population- and community- level effects. Note that few information sources include a complete suite of data (that is, toxicity data accompanied by data for concentrations of toxicants in the environment and in biota). We provide a data extraction (Rowe et al. 2002 data) to facilitate data comparison.
Rowe et al. 2002 Data (xls) (.xls file)
References
- Bartholomew JL, Lorz HV, Sollid SA, Stevens DG (2003) Susceptibility of juvenile and yearling bull trout to Myxobolus cerebralis and effects of sustained parasite challenges. Journal of Aquatic Animal Health 15:248-255.
- Beak International Incorporated (2002) Literature review of environmental toxicity of mercury, cadmium, selenium and antimony in metal mining effluents. Natural Resources Canada, The Mining Association of Canada, and Environment Canada.
- Bengtsson BE, Larsson A (1986) Vertebral deformities and physiological effects in fourhorn sculpin (Myxocephalus quadricornis) after long-term exposure to a simulated heavy metal-containing effluent. Aquatic Toxicology 9:215-229.
- Borgmann U, Couillard Y, Doyle, Dixon DG (2005) Toxicity of sixty-three metals and metalloids to Hyalella azteca at two levels of water hardness. Environmental Toxicology and Chemistry 24(3):641-652.
- Clements WH, Cherry DS, Van Hassel JH (1992) Assessment of the impact of heavy-metals on benthic communities at the Clinch River (Virginia): evaluation of an index of community sensitivity. Canadian Journal of Fisheries and Aquatic Sciences 49(8):1686-1694.
- Di Toro DM, Allen HE, Bergman HL, Meyer JS, Paquin PR, Santore RC (2001) Biotic ligand model of the acute toxicity of metals. 1. Technical basis. Environmental Toxicology and Chemistry 20(10):2383-2396.
- Hunn JB, Schnick RA (1990) Toxic substances. in: Meyer FP, Barclay LA (Eds). Field Manual for the Investigation of Fish Kills. U.S. Fish and Wildlife Service, Washington DC. Resource Publication 177.
- Kapustka LA, Clements WH, Ziccardi L, Paquin PR, Sprenger M, Wall D (2004) Issue Paper on the Ecological Effects of Metals. U.S. Environmental Protection Agency, Risk Assessment Forum.
- Langmuir DL, Chrostowski P, Vigneault B, Chaney RL (2004) Issue Paper on the Environmental Chemistry of Metals. U.S. Environmental Protection Agency, Risk Assessment Forum. 113 pp.
- Marsalek J, Jimenez-Cisneros BE, Malmquist PA, Goldenfum J, Chocat B (2006) Urban water cycle processes and interactions. UNESCO-IHP. IHP VI, Technical Documents in Hydrology No. 78. 152 pp.
- Naimo TJ (1995) A review of the effects of heavy metals on freshwater mussels. Ecotoxicology 4:341-362.
- Pollard AI, Yuan L (2006) Community response patterns: Evaluating benthic invertebrate composition in metal-polluted streams. Ecological Applications 16(2):645-655.
- Rowe CL, Hopkins WA, Congdon JS (2002) Ecotoxicological implications of aquatic disposal of coal combustion residues in the United States: a review. Environmental Monitoring and Assessment 80:207-276.
- Sippel AJA, Geraci JR, Hodson PV (1983) Histopathological and physiological responses of rainbow trout (Salmo gairdneri Richardson) to sublethal levels of lead. Water Research 17(9):1115-1118.
- Sorensen EMB (1991) Metal Poisoning in Fish. CRC Press, Boca Raton FL. 374 pp.
- Sures B (2001) The use of fish parasites as bioindicators of heavy metals in aquatic ecosystems: a review. Aquatic Ecology 35(2):245-255.
- U.S. EPA (2005) Methods/Indicators for Determining when Metals are the Cause of Biological Impairments of Rivers and Streams: Species Sensitivity Distributions and Chronic Exposure-Response Relationships from Laboratory Data. U.S. Environmental Protection Agency, Office of Research and Development, Washington DC. EPA/600/X-05/027.
Contact: Authors & Contributors